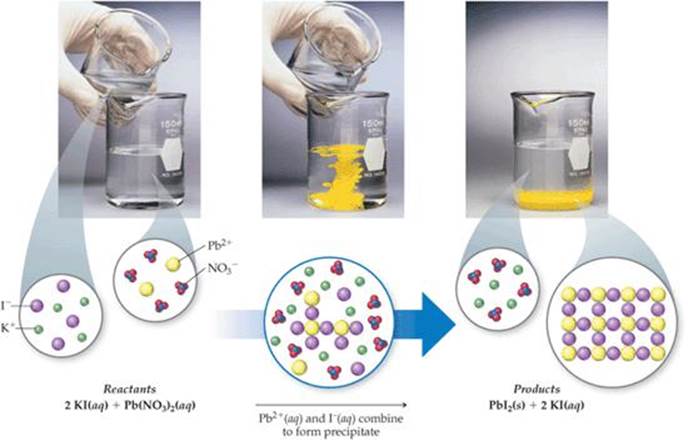 
There are many other factors that can affect solubility, but these rules are a good first step to determine the outcome of aqueous solution reactions. The solubility rules are a useful guideline to predict whether a compound will dissolve or form a precipitate. The finished reaction is:Ģ KCl(aq) + Pb(NO 3) 2(aq) → 2 KNO 3(aq) + PbCl 2(s) A precipitation reaction refers to the formation of an insoluble salt when two solutions containing soluble salts are combined. This means PbCl 2 is insoluble and form a precipitate. Chlorides are soluble in water with the exception of silver, lead and mercury. KNO 3 will remain in solution since all nitrates are soluble in water. Precipitation reactions Google Classroom About Transcript A lot of ionic compounds dissolve in water, dissociating into individual ions. The products should rearrange the ions to: What would be the expected products and will a precipitate form? The resulting balanced reaction would be:Ģ AgNO 3(aq) + MgBr 2 → 2 AgBr(s) + Mg(NO 3) 2(aq) The other compound Mg(NO 3) 2 will remain in solution because all nitrates, (NO 3) -, are soluble in water. For example, the first reaction should be those containing barium chloride. Write these net ionic reactions in alphabetical order as they appear above. The process that forms a precipitate is called precipitation. Use a solubility chart to determine the identity of the precipitate. A precipitate is an insoluble compound that is formed in a liquid solution by mixing two soluble salts together. These insoluble salts formed in precipitation reactions are called precipitates. For each combination of solutions that GAVE A PRECIPITATE, write the correct net Ionic equation. Are the products soluble in water?Īccording to the solubility rules, all silver salts are insoluble in water with the exception of silver nitrate, silver acetate and silver sulfate. The term ‘precipitation reaction’ can be defined as a chemical reaction occurring in an aqueous solution where two ionic bonds combine, resulting in the formation of an insoluble salt. The state of the products needs to be determined. The balanced reaction would be:Ģ AgNO 3(aq) + MgBr 2 → 2 AgBr(?) + Mg(NO 3) 2(?) For example, a silver nitrate solution (AgNO 3) is mixed with a solution of magnesium bromide (MgBr 2). 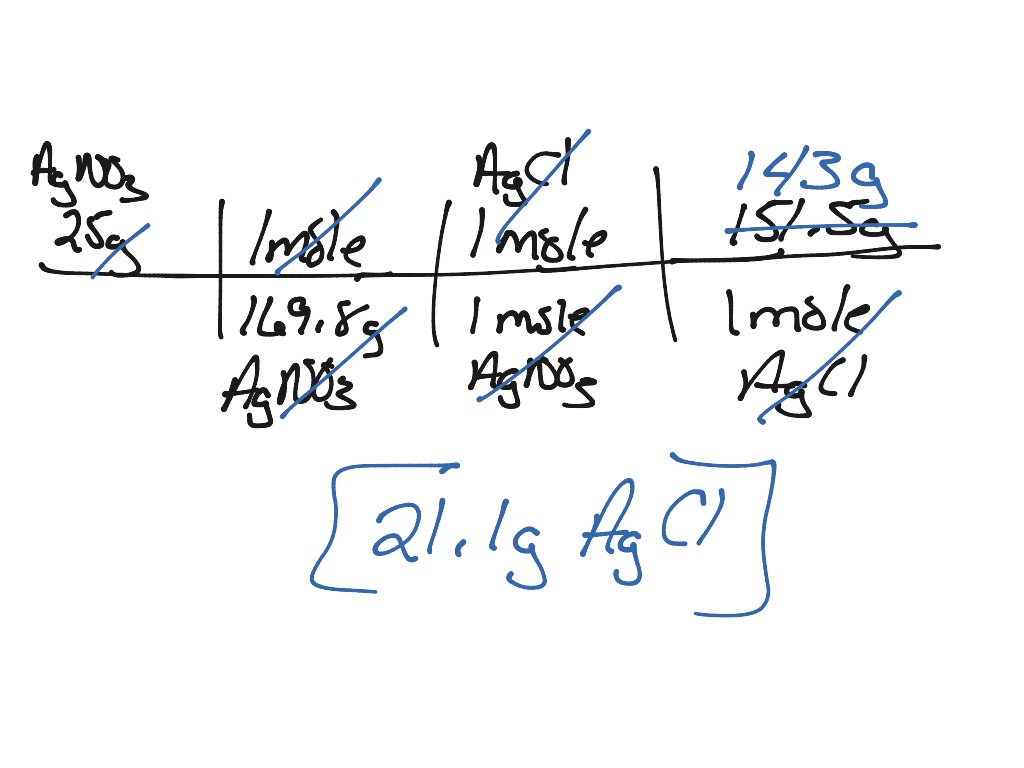
The question remains, will AD or CB remain in solution or form a solid precipitate?Ī precipitate will form if the resulting compound is insoluble in water. This reaction is generally a double replacement reaction in the form: When two aqueous solutions are mixed, the ions interact to form products. These solutions are represented in chemical equations in the form: AB(aq) where A is the cation and B is the anion. This guide will show how to use the solubility rules for inorganic compounds to predict whether or not the product will remain in solution or form a precipitate.Īqueous solutions of ionic compounds are comprised of the ions making up the compound dissociated in water. When two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a solid precipitate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
